Part 7: Screening and Surveillance to Identify Hospital-acquired Infection
- Introduction
- Policies for Screening Patients and Staff to Identify Infection
- What Is Surveillance?
- What Surveillance Is Undertaken for Hospital-acquired Infection?
- Infection Control Teams’ Access to Surveillance Data
- Reporting Surveillance Results
- Surveillance of Patients After Discharge from Hospital
- Assessing the Need for Isolation Facilities
Introduction
7.1
In this part we consider how hospital services identify infections by:
- “screening” – which involves taking swabs or blood samples, and undertaking tests to determine whether a person is colonised or infected; and
- “surveillance” – which involves the collection and interpretation of data on infections, and reporting the results so that clinical staff and managers can take action, if appropriate.
7.2
Isolation facilities may be required for patients identified as having a transmissible infection or as being particularly vulnerable to infection. In this part, we also consider hospital services’ arrangements for isolating patients.
Policies for Screening Patients and Staff to Identify Infection
7.3
When colonised or infected patients and/or staff are identified, action can be taken to help prevent the spread of infection and, if appropriate, begin any necessary treatment.
7.4
In August 2002, the Ministry published guidelines for the control of Methicillin-resistant Staphylococcus aureus (MRSA). These guidelines suggest when patients and staff should be screened, and explain what steps should be taken if a person is found to have acquired MRSA. The reasons why it is important to control MRSA are set out in Figure 15 (on the next page).
Figure 15
Why Is It Important to Control Methicillin-resistant Staphylococcus aureus (MRSA)?
| Why Is It Important to Control Methicillin-resistant Staphylococcus aureus (MRSA)? |
|---|
| MRSA is resistant to methicillin, oxacillin, and other antibotics. It can spread readily in hospitals and other health care settings, primarily through direct person-to-person contact. In some people, MRSA may become invasive and cause disease. Most vulnerable are those who are ill or injured. It can cause a wide range of infections, including skin abscesses, post-operative wound infections, septicaemia, and pneumonia. Because of their resistance to antibiotics, infections caused by MRSA have become increasingly difficult to control, requiring more complicated, toxic, and expensive treatment. MRSA can survive for long periods and, once established in a hospital or other health care setting, can be difficult to eradicate. |
7.5
Screening for infectious organisms may involve testing staff and/or patients (with their consent):
- who have been admitted with infections resulting from other organisms;
- after an outbreak, when the source is being investigated;
- as a precautionary measure, when patients are transferred from one hospital to another; and
- when staff have been exposed to an infected patient’s blood or other body-fluids.
7.6
Staff colonised with an infectious organism could potentially infect others, and should be screened before taking up their position. Summary information from screening can be used to:
- establish baseline test results against which to measure exposure in the future; and
- assess the need for and cost-effectiveness of a staff vaccination programme.
7.7
We asked:
- whether the hospital service had a routine screening programme, who was responsible for screening, and whether the programme included both staff and patients; and
- what the programme involved.
7.8
We present the survey responses for the screening of patients and the screening of staff separately in the following paragraphs.
Survey Responses on Screening Patients
7.9
Twenty hospital services had a routine screening programme for certain groups of patients.183
7.10
Infectious organisms such as MRSA are known to be more prevalent in some health care settings than others. Directing preventive strategies (including screening) at patients admitted to hospital from high-risk settings can be cost-effective by reducing rates of hospital-acquired infection and use of antibiotics, and the length and cost of the time patients spend in hospital.
7.11
One hospital that we visited set out to establish whether there was a difference in MRSA colonisation rates between patients admitted to hospital from health care facilities and those admitted from the wider community (generally from their own home). Patients from selected health care facilities were screened, along with patients from the wider community.
7.12
The results of this screening identified key risk factors for MRSA colonisation – including patient characteristics, medical history, and residence. For example, patients admitted from aged-care facilities were shown to be relatively more likely to be colonised with MRSA. Such information on risk factors can be used to make the hospital’s screening programme more effective by targeting higher-risk patients for screening on admission to the hospital.
7.13
Hospitals can usefully screen patients for a number of multi-drugresistant organisms on admission. A tertiary hospital that we visited required certain patients to be screened for three organisms, as shown in Figure 16 (on the next page). Another hospital service was screening all patients aged over 65 who had been in hospital for a week or more, and was achieving earlier detection of multi-drug-resistant organisms as a result.
Figure 16
Protocol for Screening Patients on Admission for Multi-drug-resistant Organisms
| Patient’s Profile | ESBL184 | MRSA | VRE185 |
|---|---|---|---|
| From rest home/long-term care facility. | X | ||
| From another District Health Board hospital. | X | ||
| Previous admission to xxxx or yyyy hospital in the last two years. | X | ||
| Inpatient in zzzz hospital any time since January 2000. | X | ||
| Admission to any overseas hospital in the last six months. | X | X | X |
| X = screen on admission | |||
Survey Responses on Screening Staff
7.14
All hospital services carried out pre-employment screening for MRSA for clinical staff, and many also screened new staff for other infections such as Hepatitis B and TB.186 Sixteen hospital services routinely screened staff to check whether any were colonised or infected.187 This screening:
- confirms employees are free from being colonised or infected;
- indicates where staff (particularly those with blood-borne diseases) can safely work in a hospital, minimising risks to patients; and
- provides baseline information so that future exposures to infectious diseases can more easily be identified.
| "… These outbreaks were all caused by strains introduced into the hospital from overseas. Consequently, there is an active, ongoing staff MRSA screening programme and surveillance of patients being admitted from overseas hospitals …" |
7.15
Hospital services need to be especially alert to the risks associated with staff who have worked in other hospitals or institutions (including overseas) that have a high incidence of antibiotic-resistant infection. The Ministry’s MRSA guidelines and the TB guidelines188 contain advice on the circumstances in which screening is appropriate – including criteria for screening staff. For example, the guidelines recommend that one set of negative swabs be obtained before staff previously exposed to MRSA are cleared for general duties. Where a staff member was previously found to be colonised or infected with MRSA, three consecutive sets of negative swabs are required.
7.16
Tracing and screening is required in the event of an infection outbreak. For example, in one hospital that we visited TB had been unexpectedly found in a patient’s wound. A total of 46 staff were identified as having possibly been exposed to the infection through:
- direct exposure to the patient (usually in dressing the wound);
- care of the patient without direct exposure to the wound; and
- indirect contact without physical or clinical involvement.
7.17
The importance of exhaustive tracing and screening was demonstrated when three of the screened staff showed signs of infection.
7.18
Our survey showed that responsibilities for screening staff and patients differed:
- In 10 hospital services, responsibility for screening patients rested with the infection control team.189
- In the other hospital services, ward staff or the admitting doctor or nurse carried out patient screening.190
- Staff were, in most cases, screened by occupational health or human resources personnel.191
Conclusions
7.19
Screening patients and staff plays an important part in preventing and controlling infectious diseases in a hospital. Most hospital services undertake targeted screening of patients and screening of staff. Those that do not, run a relatively higher risk of infections entering their hospital.
7.20
Exhaustive tracing and subsequent screening of staff and patients are essential responses after an outbreak of a hospital-acquired infection, in order to contain the further spread of the infection.
What Is Surveillance?
7.21
The Standard defines surveillance as:
... the regular collection, collation and analysis of information on infection events and rates, either continuously or at regular intervals, and the timely dissemination and feedback to those who need to know.192
7.22
Surveillance is designed to:
- monitor patterns of infectious disease (including their causes), enabling preventive and control measures to be put in place;
- prevent outbreaks or detect them early in order to initiate timely action;
- identify groups of patients at risk of disease; and
- provide information for planning of infection control services and allocation of resources.
7.23
Research193 has shown that surveillance can substantially reduce levels of hospital-acquired infection. In the following paragraphs we examine:
- what surveillance hospital services are undertaking for hospital-acquired infection;
- what access infection control teams have to surveillance data;
- reporting of surveillance results; and
- the surveillance of patients after discharge from hospital.
What Surveillance Is Undertaken for Hospital-acquired Infection?
7.24
We expected all infection control teams to be carrying out surveillance activities as part of their infection control programmes. Surveillance should:
- use common definitions that make it possible to collate surveillance data from hospitals and enable valid comparisons to be made;
- focus on a range of factors including organisms, cases, wards or units, and infection sites (e.g. surgical wound infection, bloodstream infection, etc.);
- draw on all data sources likely to contain information about infections, their causes and relevant circumstances; and
- produce results that are shared with appropriate clinical staff to support discussion of the improvements required in clinical and other hospital practice.
Survey Responses on Common Surveillance Data Definitions
7.25
Collaboration within the hospital on the collection and analysis of infection control data can generate useful surveillance results. To do this properly, it is essential that staff agree and clearly understand common data definitions.
7.26
In one hospital that we visited, the infection control nurse reviewed all positive blood cultures for evidence of bloodstream infection. The clinical microbiologist considered the results of these reviews each month. Total bloodstream infections over quarterly periods were converted into annual rates for 1000 inpatient admissions. The analysis revealed the frequency of different infection sites (such as surgical wounds and sites of intravenous catheters)194, the organisms causing such episodes, and the patient groups most affected. Critical to the quality and reliability of this analysis was the use of a large sample of consistent data taken from throughout the hospital.
7.27
It is important to have agreed definitions for surveillance data, so that the data collected from different sources and by different people can be readily interpreted and compared. Our survey found that hospital services were using a variety of national and international definitions195, with some using more than one set of definitions. This makes it impossible to interpret and compare data within or between DHBs or hospital services.
Survey Responses on the Focus of Surveillance
7.28
All infection control teams reported that they were carrying out some form of surveillance.196 Surveillance should look at a range of events. The Standard provides guidance on the possible types of surveillance activities, and identifies the following events that are typically monitored:
- surgical site infections;
- bloodstream infections;
- pneumonias;
- device-related infections; and
- infections resulting from multi-drug-resistant organisms.
7.29
The focus of surveillance will differ according to the size of the health facility. The Standard specifies types of events that all hospital facilities should target, and suggests that post-discharge surveillance should be undertaken for some specific events such as surgical site infections. Larger facilities would generally focus on specific high-risk areas, while smaller facilities would look at all events throughout the organisation.
7.30
We asked hospital services whether they had carried out surveillance using any of the following four methods during the 12 months to June 2001:
- Continuous alert organism surveillance – this involves reviewing the results of laboratory tests for the presence of significant organisms197 as an indicator of the status of infection in the hospital.
- Continuous alert condition surveillance – the same broad approach but focused on those groups of patients most likely to acquire an infection or who are particularly vulnerable should they acquire an infection. This method requires close liaison between the infection control team and ward staff, and relies on ward staff being able to identify those patients who should be monitored, and to recognise an infection from the patient’s condition.
- Targeted surveillance – this is similar to the first two methods, but is undertaken periodically rather than continuously. It involves surveillance of patients according to certain characteristics – within a particular case mix, in a specific ward or unit, or by site of infection.
- Selective laboratory-based ward liaison surveillance – using this method, laboratory and ward staff review selected surveillance data. This method can reveal an increase in the incidence of infection in a health care setting before it becomes a problem.
7.31
During the 12 months to June 2001, 19 infection control teams had undertaken continuous alert organism surveillance.198 Eleven infection control teams had complemented this approach with the second method – continuous alert condition surveillance.199
7.32
All hospital services collect information on incidences of hospital-acquired bloodstream infection for the Ministry’s Balanced Scorecard reporting (see paragraph 2.54 on page 51). All but two respondents undertook some form of targeted surveillance (method 3) focused on specific sites of infections (e.g. wounds) and/or wards or units of the hospital.200 Twelve had used the collaborative, laboratory-based ward liaison surveillance method.
7.33
Of the six hospital services with tertiary hospitals, only three reported that they were undertaking some form of surgical site surveillance.201 Only one had a comprehensive programme for the surveillance and monitoring of hospital-acquired infection that encompassed all of the following:
- overall rates of infection and rates from twice-yearly prevalence studies202;
- infectious organisms;
- common infection types such as those in the bloodstream and surgical wounds; and
- cases associated with particular surgical procedures.
Conclusions
7.34
Surveillance is an essential component in the prevention and control of infection in hospitals. All hospital services undertake some form of surveillance and, for most, this involves a programme for monitoring laboratory samples for significant organisms.
7.35
It is difficult to use surveillance data to draw meaningful comparisons within and between hospital services – or to collate national and regional infection data for comparable facilities undertaking similar medical procedures – because hospital services do not use consistent data definitions.
7.36
Hospital services with tertiary hospitals need to manage the additional infection risks associated with complex surgery and the relatively high severity of illness of many of their patients. However, only one of these six hospital services had a comprehensive programme of surveillance, so some important dimensions are not being covered.
| Recommendation 35 |
|---|
| In consultation with DHBs, the Ministry should draw up guidance on how and to what extent surveillance data should be collected. |
Infection Control Teams’ Access to Surveillance Data
7.37
Infection control teams need ready access to surveillance data in order to monitor infection rates and trends. Such data might include:
- details of patients with infections and patients most at risk of infection; and
- information about organisms, and laboratory results containing information about disease-resistance patterns.
7.38
Systems should be designed to alert staff to issues such as the presence of multi-drug-resistant organisms and of patients re-admitted with an infection.
7.39
Efficient systems for collecting and recording data are particularly important in larger hospitals where patient throughput is high, hospital systems are more complex, and where it would otherwise be very difficult to maintain oversight of hospital-acquired infection. Where relevant hospital information systems – such as electronic patient records, laboratory, and pharmacy systems – are linked and readily accessible, they can provide infection control teams with important information on trends in hospital-acquired infection throughout the hospital.
7.40
The infection control team in one hospital that we visited had good access to surveillance data and was able to analyse bloodstream infections to identify and target vulnerable patient groups, and to improve the way in which medical procedures were performed. As indicated in paragraph 7.26 on page 159, the analysis provided a range of useful results, including:
- those services within the hospital responsible for the highest incidence of bloodstream infection;
- those medical and surgical procedures representing the main sources of infection (such as bone marrow transplants and intravenous procedures); and
- the organisms responsible for infection.
7.41
A more detailed analysis of infections that occurred after intravenous procedures showed which wards, methods of intravenous procedure, and organisms were most commonly associated with these infections.
Survey Responses on Sources of Surveillance Data
7.42
Most infection control practitioners and doctors had full or partial access to electronic patient databases, the reports of microbiological analysis, and the hospital intranet.203
7.43
The infection control teams drew surveillance data from the following sources:204
- laboratory reports – the most commonly used source – 16 infection control teams examined these daily, three examined the results weekly, and one examined them monthly;
- electronic patient records system – 17 teams examined these daily (12) or weekly (5);
- patients’ paper records – 16 examined these daily (6) or weekly (10).
7.44
Only six infection control teams examined pharmacy prescribing reporting systems when carrying out surveillance activities.205 The infection control team had full or partial access to the pharmacy prescribing system in only four hospital services.206 Lack of access for infection control teams may impede their ability to work with the pharmacy to identify relationships between patterns of antibiotic prescribing and infections.
7.45
Data on infection rates was most commonly collected by organism, by site of infection (e.g. wounds), and by ward or unit. Only eight teams collected data by risk factor (e.g. the patient’s condition and/or whether they were subject to an invasive procedure) and 11 teams collected data by surgeon.207 Collecting or collating data in this way would enhance analysis and improve targeting to address possible causes of infection.
Conclusions
7.46
Overall, most infection control teams have satisfactory access to information systems for surveillance purposes. Most teams undertake regular and timely examination of surveillance data – mainly laboratory reports and electronic patient records.
7.47
The data is collected so that it can be analysed against a range of factors, though not always by risk factor or by surgeon. In those hospital services collecting this additional data, a more extensive analysis is possible to help target causes of infection.
Reporting Surveillance Results
7.48
Where a patient’s specimen indicates the presence of an infection, the responsible clinician receives a written report of the results.
7.49
Collating these results – for example, by case mix, ward, or site of infection – and making them available to clinical staff helps identify patterns of infection. Analysing and providing the results to clinical staff enables them to address identified concerns, take remedial action, and change practices that might be allowing infection to start or spread (see example provided in Figure 17 below).
Figure 17
Example of Analysis of Infection Data that Can Produce Valuable Results for Clinicians
| Example of Analysis of Infection Data that Can Produce Valuable Results for Clinicians |
|---|
| Surgical site infections are a common hospital-acquired infection, resulting in sickness or, very occasionally, death. Patients whose surgical wounds become infected may need to spend additional days in hospital for treatment and rehabilitation. Monitoring and analysis of infection rates, with feedback to clinicians to help them review their practice, can help prevent such infections. The details below illustrate one hospital’s plan for analysing rates of surgical site infection over a specific period. Surveillance Period: 133 days. Information to be collected: • Admission/operation date. • Discharge date. • Surgeon. • Length of surgery. • Type of surgery. Calculation: Surgical risk scores for patients. Monitoring: Patient health after discharge from the hospital. Summary of results – examples of the questions posed: • Was the infection identified in hospital or after discharge? • What is the relationship between risk scores and levels of infection? • What organisms have been responsible for infections? • What have been the outcomes for the affected patient groups? |
Survey Responses on Reporting of Surveillance Results
7.50
Twenty infection control teams reported infection rates, and 12 reported trends.208 Both measures are necessary to provide a complete current and historical picture.
7.51
Not all hospital services reported the results of surveillance activities to clinical staff. Only two-thirds of hospital services that analysed surveillance results by ward/unit and by site of infection during the year to June 2001 reported the results to medical or nursing staff.209
7.52
Most hospital services were periodically reporting surveillance results to infection control committees and to key clinical managers:210
- twenty infection control teams reported the results to the infection control committee at least annually, with 16 doing so monthly or more often; and
- eighteen Medical Directors and Directors of Nursing received surveillance results at least annually, and more than half at least monthly.
7.53
In four cases, quality/risk managers received no surveillance reports.211
Conclusions
7.54
Reporting of surveillance data to clinical staff needs to be selective in order to avoid burdening them with non-essential information. However, some infection control teams were not reporting this data to them at all – thereby losing the opportunity to support clinical staff in exploring possible causes of infection, such as clinical practices, that might be affecting the level of infection.
7.55
Not all infection control teams were providing periodic surveillance reports to quality/risk managers, highlighting (as noted elsewhere in this report) the poor links in some hospital services between infection control activities and the roles of quality assurance and risk management.
| Recommendation 36 |
|---|
| Infection control teams should review how reporting of surveillance data to quality/risk managers and clinical staff can be improved. |
Surveillance of Patients After Discharge from Hospital
7.56
Surgical patients are three times more likely than other patients to acquire an infection in hospital. Many hospital-acquired infections become evident only after a patient has been discharged.
Survey Responses on Patients Re-admitted with a Hospital-acquired Infection
7.57
The most serious infections may lead to patients being re-admitted, so the collation of information on patients re-admitted with a hospitalacquired infection is an important part of infection control. However, only seven hospital services were able to identify patients re-admitted with a hospital-acquired infection from an electronic patient administration system212 and, even in those cases, the information was not always considered reliable.213
Survey Responses on Post-discharge Surveillance
7.58
Most hospital-acquired infections that become apparent after discharge are not serious enough to require re-admission, but they can still cause the patient discomfort and lead to additional health care costs. Such patients are often treated in the community without the knowledge of the hospital. As a consequence, reported infection rates are likely to be understated.
7.59
An investigation into the health of discharged patients by the infection control team at one large hospital we visited, illustrated the extent to which hospital-acquired infection is likely to be understated, and demonstrated the benefits to be gained from post-discharge surveillance.
7.60
For the period March to April 2001, the infection control team had collected information about 237 surgical operations. As part of its investigation, the team analysed records of re-admissions and responses to a questionnaire sent to patients 30 days after discharge. The team found that surgical wound infections had occurred in 18 patients (7.6%), with all but two having occurred after discharge.
7.61
Comprehensive monitoring of patient health after discharge would be time-consuming and costly, but we expected all hospital services to be carrying out some form of selective post-discharge surveillance. We asked whether the infection control team carried out post-discharge surveillance and how it identified cases of hospital-acquired infection.
7.62
Sixteen infection control teams carried out some form of post-discharge surveillance. Fourteen of these teams asked patients to fill in a questionnaire. Five used telephone surveys of patients and two surveyed the patients’ general practitioners (GPs).214
7.63
Patients who acquire infections as a result of their stay in hospital are usually treated by their GPs. The Ministry plans to enhance sharing of data between GPs and other community health facilities, pharmacies, and hospitals.215 Enhanced data-sharing has the potential to provide hospitals with a more accurate picture of their rates of hospital-acquired infection.
Conclusions
7.64
It is important to know when patients are re-admitted with a hospitalacquired infection because these are generally the most serious cases, but most hospital services have no reliable information on such re-admissions.
7.65
Comprehensive monitoring of every patient after discharge (including patients who attend as outpatients or day cases) is unlikely to be costeffective at present. However, most hospital services undertake periodic post-discharge surveillance which provides an indication of the extent of hospital-acquired infection that becomes apparent after discharge.
| Recommendation 37 |
|---|
| Hospital services should put in place systems to help them identify patients re-admitted with a hospital-acquired infection. |
Assessing the Need for Isolation Facilities
7.66
Patients with infectious conditions can pass those conditions to other patients, staff, and other people within the hospital. If a patient is known (or suspected) to be colonised with a particular infection, they may need to be physically isolated from other people. The design of isolation rooms (for example, whether rooms have a specialised ventilation system or their own wash basin and toilet) should have regard to the way in which the particular infection can be transmitted (for example, by body contact or by air) and the condition of the patient.
| "… Too few toilets and showers in wards therefore isolated patients required to shower last to enable showers to easily be decontaminated afterwards … " |
7.67
Protective isolation facilities may also be necessary for patients who have no immunity or whose immunity is impaired, such as those undergoing certain types of medical procedures (e.g. bone marrow transplants).
Survey Responses on Arrangements for Isolating Patients
7.68
Hospitals need to have enough appropriately located and designed isolation rooms to meet their requirements. We asked infection control teams:
- whether hospital services had undertaken a formal risk assessment to ensure that adequate arrangements were in place to isolate patients; and
- whether the team assessed isolation arrangements as satisfactory.
7.69
Fewer than half of the 21 hospital services (including two of the six with tertiary hospitals) had undertaken a formal written risk assessment to ensure that adequate arrangements were in place for isolating patients.216 Most risk assessments considered what type of rooms were needed and how many.217
7.70
Only 12 infection control teams viewed the hospital service’s arrangements for isolating patients with transmissible diseases218 as very or fairly satisfactory (see Figure 18). Only 13 infection control teams viewed the hospital service’s arrangements for protective isolation of patients with low immunity219 as very or fairly satisfactory.
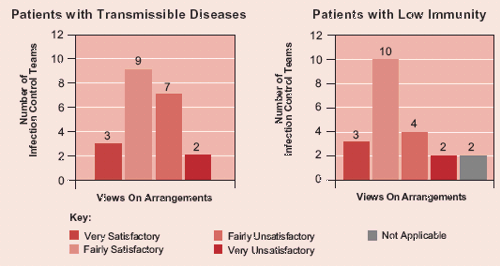
Figure 18
Infection Control Teams’ Views on the Arrangements for Isolating:
• Patients with Transmissible Diseases
• Patients with Low Immunity
7.71
Where respondents assessed the facilities as fairly or very unsatisfactory they gave a range of reasons:
- a lack of adequate en-suite facilities;
- not enough suitably ventilated rooms;
- rooms that were too small;
- rooms that were difficult to clean; and
- facilities that did not comply with the guidelines for the care of patients with TB.
7.72
Isolation facilities are a key resource in tertiary hospitals which carry out many complex surgical procedures, treat especially sick and vulnerable patients, and may have high rates of bed occupancy. Some infection control staff in the tertiary hospitals we visited were worried by the demand on bed availability, and shortages of rooms with specially designed ventilation systems for patients with transmissible diseases.
| "… Demand for isolation rooms frequently exceeds availability …" |
7.73
One team in a hospital service with a tertiary hospital viewed the isolation arrangements – for both transmissible diseases and protective isolation of patients with low immunity – as very unsatisfactory.220 Concern was expressed at the lack of specialist arrangements for isolating patients undergoing bone marrow transplants.
7.74
Isolation rooms should be located close to the wards where the relevant patients are undergoing treatment and near the specialists and other staff providing that treatment. One hospital we visited had a specially designed room for patients with multi-drug-resistant TB, but the room was poorly placed for the staff providing the treatment. As a result, TB patients were being admitted to a respiratory ward located in a different part of the hospital. This practice had the potential to place other patients at risk and meant that the special room was not being used for its intended purpose.
7.75
Treating patients with low immunity demands special precautions. In the same hospital service’s haematology unit (which treats patients with blood cancers like leukaemia) a ventilation system minimised the risks of infection to this particularly vulnerable group of patients. The air in the unit was tested monthly for fungal spores, helping to maintain a safe environment.
Conclusions
7.76
We were concerned that most hospital services were not undertaking formal risk assessments of the adequacy of arrangements for isolating patients.
7.77
In some hospital services, the insufficient quality and/or quantity of isolation facilities places some patients at risk from having to undergo and/or recover from complex technical and expensive treatment in a poor quality environment that is not suited to its purpose.
| Recommendation 38 |
|---|
| Hospital services should review the adequacy of their arrangements for isolating patients. |
183: F3.3: Q42, c, d.
184: Extended spectrum Beta lactamase.
185: Vancomycin-resistant enterococci.
186: F3.3: Q45, B, C.
187: F3.3: Q42, a, b; Q45, A.
188: Guidelines for the Control of Methicillin-resistant Staphylococcus aureus (MRSA) in New Zealand, Ministry of Health, August 2002; Guidelines for Tuberculosis Control in New Zealand, Department of Health, 1992.
189: F3.3: Q43, b.
190: F3.3: Q43, b; Q44.
191: F3.3: Q43, a, Q44.
192: Appendix A of NZS 8142:2000 Infection Control.
193: For example: Haley et al The efficacy of infection surveillance and control programs in preventing nosocomial infections in US hospitals; American Journal of Epidemiology 1985; Vol. 121: pages 182-205. (A study of 338 hospitals, which found that hospitals with active surveillance and control programmes reduced the incidence of nosocomial infection by 32% over five years; those without had an increase of 18%). Cruse P Surgical wound sepsis; Canadian Medical Association Journal 1970; Vol. 102: pages 251-258. Chelgren G, Laforce FM Limited, periodic surveillance proves practical and effective Hospitals 1978; Vol. 52: pages 151-154.
194: A catheter may, for example, be inserted into the skin so that a tube can be attached leading to a bag of intravenous fluid – the place where the catheter is inserted is referred to as the “site”.
195: F3.3: Q13a & Q13b.
196: F3.3: Q12.
197: “Significant organisms” are those organisms that the hospital’s experienced clinicians and laboratory personnel consider necessary to monitor.
198: F3.3: Q14, A – a.
199: F3.3: Q14, A – b.
200: F3.3: Q14, A – d, e.
201: F3.3: Q14, A – e.
202: A prevalence study examines the extent to which a phenomenon (in this case an infection) is present at a particular time in a particular area.
203: F3.1: Q37, a, b, d.
204: F3.3: Q16.
205: F3.3: Q16, d.
206: F3.1: Q37, c.
207: F3.3: Q17.
208: F3.3: Q18, a & b respectively.
209: F3.3: Q14, B & C – d, e.
210: F3.3: Q21.
211: F3.3: Q21, e.
212: F3.1: Q38.
213: Some respondents’ additional notes to F3.1: Q38.
214: F3.3: Q22.
215: Ministry of Health, The WAVE Advisory Board to the Director-General of Health, 2001, From Strategy to Reality. The WAVE Project Kia hopu te ngaru – Health Information Management and Technology Plan – Working to Add Value through E-information, Ministry of Health, Wellington,– www.moh.govt.nz.
216: F3.3: Q46, a & b.
217: F3.3: Q47, a & b.
218: F3.3: Q51a.
219: F3.3: Q51b.
220: F3.3: Q51a; Q52a.
page top