Part 2: The Framework for Infection Control
- Introduction
- New Zealand Public Health and Disability Act 2000
- Health and Disability Services (Safety) Act 2001
- Using Voluntary Accreditation to Prepare for Certification
- Promoting Health and Disability Standards
- Using Expert Advisory Groups
- Undertaking National Surveillance
Introduction
2.1
Infection control in public hospitals sits within an overall framework for ensuring the quality of care provided to patients – in terms of safety, effectiveness, and patient satisfaction.
2.2
In this part of the report, we:
- examine the framework for infection control in the context of quality and risk management;
- set out the main statutory requirements, including the roles and obligations of the Ministry and DHBs, with specific reference to infection control; and
- discuss the two key pieces of legislation that establish the framework governing the quality of public health care–
- the Public Health and Disability Act 2000; and
- the Health and Disability Services (Safety) Act 2001.
2.3
Figure 2 (on the next page) shows how these Acts relate to the overarching role of the Ministry and the role of DHBs in directly ensuring the provision of public health care services.
Figure 2
Responsibilities for the Provision of Public Health Care Services

2.4
We then examine the role of the Ministry in:
- encouraging DHB hospital services to pursue voluntary accreditation as one means of preparing for certification under the Health and Disability Services (Safety) Act 2001;
- promoting the implementation of quality standards;
- using existing and specific-purpose expert groups to provide authoritative advice on clinical issues; and
- undertaking national surveillance of hospital-acquired infection.
New Zealand Public Health and Disability Act 2000
2.5
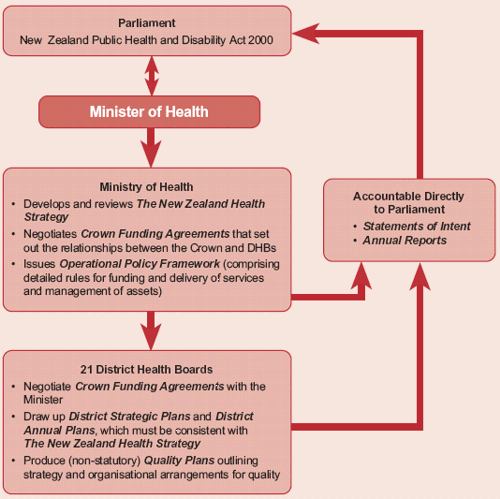
The New Zealand Public Health and Disability Act 2000 established the 21 DHBs from 1 January 2001, and defined their roles and functions. Figure 3 opposite illustrates the key accountability documents required by the Act, including the requirement for the Minister to establish a strategy for the delivery of health care services.28 The Act also requires the Minister to determine strategies for the preparation and use of standards, quality assurance programmes, and performance monitoring to promote patient safety.
Figure 3
Accountability under the New Zealand Public Health and Disability Act 2000
The New Zealand Health Strategy and the Strategic Plans of District Health Boards
2.6
Quality is a key focus of The New Zealand Health Strategy (the Strategy); one of its seven principles is a high performing system in which people have confidence.29 The Strategy:
- is designed to foster a culture that supports continual quality improvement in the delivery of services; and
- envisages reducing preventable harm – through using a risk management approach and fostering consistency of practice through shared learning, benchmarking, and clinical governance within a framework of standards.
2.7
The Strategy notes that:
... services are deemed safe when components and systems are in place that reduce risk to levels which are considered acceptable according to recognised and, where appropriate, international standards.
2.8
DHBs must reflect the Strategy in their District Strategic Plans and District Annual Plans, which identify their objectives and explain how they will meet them in their local areas. Ministry guidelines for the plans’ format and content include quality and risk management activities that are designed to ensure the safety of the hospital environment, including infection control.
An Integrated Approach to Infectious Disease
2.9
The Strategy lists 10 goals, including Better physical health. One objective supporting that goal is:
To reduce the incidence and impact of infectious diseases.30
2.10
In November 2001, the Ministry published a strategy entitled An Integrated Approach to Infectious Disease: Priorities for Action 2002-2006 (IAID) as one of a number of supporting strategies designed to give effect to the Strategy. The IAID defines the priorities for reducing the incidence and impact of infectious diseases and forms the basis for the Ministry’s work programme for infectious disease control.
2.11
Figure 4 below illustrates the Ministry’s plans for addressing two of the IAID’s six highest priority disease groupings that are directly relevant to the management of hospital-acquired infection.
Figure 4
Ministry Plans for Addressing Two High-priority Objectives for Infectious Disease Control
| Objectives | |
|---|---|
| • reduce transmission of blood-borne infections | • prevent and control hospital-acquired infections and antibiotic resistance |
| Actions | |
| • implement infection control policies and programmes • produce and disseminate protocols for health care workers • ensure preventive treatment where possible exposure to infection has occurred | • promote appropriate use of antibiotics • develop national guidelines for MRSA • improve national surveillance of antibiotic resistance • promote interaction between sectors (e.g. with the Ministry of Agriculture and Forestry) |
| Targets | |
| • all health institutions adopt and use the Infection Control Standard | • reduce inappropriate use of antibiotics |
Crown Funding Agreements
2.12
As indicated in Figure 3 on page 39, Crown Funding Agreements govern the accountability relationship between the Crown and each DHB. They impose obligations on DHBs, including to:
- monitor service obligations;
- follow best governance practice;
- ensure operational plans are consistent with the Strategy; and
- report quarterly to the Ministry on aspects of financial and non-financial performance.
2.13
As Crown entities under the Public Finance Act 1989, DHBs must prepare a statement of intent and annual financial statements for reporting to Parliament.
2.14
Each DHB must also:
- develop, document, implement and evaluate a transparent system for managing and improving the quality of Services to achieve the best outcomes for consumers31; and
- have a written quality plan outlining a clear quality strategy and identifying the organisational arrangements to implement the plan. The quality plan must be reviewed at least annually.
Health and Disability Services (Safety) Act 2001
2.15
At present, hospitals are licensed to deliver health care services under the Hospitals Act 1957 and the Hospitals Regulations 1993. Hospitals must maintain a safe environment and prevent the spread of communicable diseases through the use of appropriate equipment and careful clinical practice. The Ministry may inspect any licensed premises and close down any considered unsafe for patients.
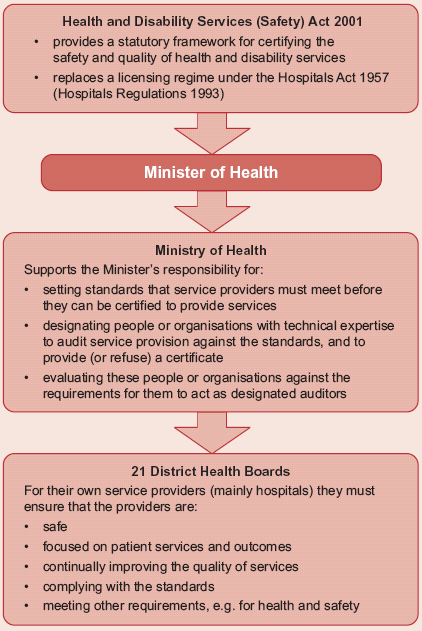
2.16
The current licensing legislation is aimed mainly at building requirements. The Health and Disability Services (Safety) Act 2001 repealed sections of the Hospitals Act related to licensing, and introduced a certification regime based on compliance with health care standards (see Figure 5 opposite). The stated purposes of the Health and Disability Services (Safety) Act 2001 are to:
- promote the safe provision of health and disability services;
- enable the establishment of consistent and reasonable standards for providing health and disability services to the public safely;
- encourage providers of health and disability services to take responsibility for providing those services to the public safely; and
- encourage providers of health and disability services to the public to improve continuously the quality of those services.32
Figure 5
Responsibilities for Implementing the Health and Disability Services (Safety) Act 2001
2.17
Under the Health and Disability Services (Safety) Act 2001, a person providing health care services of any kind must be certified by the Director-General33 of Health, and meet all relevant service standards. The legislation gives the Minister power to approve service standards, which may take a number of forms, including:
- general statements of appropriate care delivery outcomes, procedures or techniques for providing health or disability services;
- the means of achieving such outcomes; and
- technical recommendations or specifications for service delivery, or for equipment, facilities, goods or materials used in delivering services.34
2.18
The Health and Disability Services (Safety) Act 2001 requires all hospital, rest home, and disability residential care services to be audited by a designated audit agency and certified by the Director-General of Health by 1 October 2004. Service providers must meet specified standards before they can be certified.
2.19
Certification will be valid for varying periods, but usually for three years. New providers of hospital and health care services had to be certified by 1 October 2002. Providers licensed under the current arrangements (see paragraph 2.15) have a two-year transition period (1 October 2002 to 1 October 2004) to achieve certification.
2.20
The Ministry has taken steps to facilitate the transition to certification by:
- encouraging DHB hospital services to pursue voluntary accreditation as one means of preparing for the transition;
- promoting the implementation of health and disability standards; and
- using existing and specific-purpose expert advisory groups to provide authoritative consideration of clinical issues.
Using Voluntary Accreditation to Prepare for Certification
2.21
Accreditation is a voluntary way for hospital and health care service providers to demonstrate adherence to specific standards of good practice. It is entirely separate from certification under the Health and Disability Services (Safety) Act 2001, but can be used to help prepare for certification where the two sets of requirements are aligned.
2.22
When a service provider decides to obtain voluntary accreditation, it agrees to submit to assessments by clinical and health management surveyors who are trained to:
- evaluate and report on the service provider’s compliance with the accreditation standards;
- identify the service provider’s strengths and areas for improvement; and
- award accreditation if appropriate.
| "… [Accreditation] was one of the driving factors to changes in service delivery. Has assisted in improving the profile and recognition of importance of infection control for the organisation …" |
2.23
The Ministry has encouraged DHBs to seek accreditation of their hospital services to help prepare for certification, and most hospital services were taking part in an accreditation scheme that included infection control (see paragraph 4.73 on page 95). More generally, the Ministry is also promoting accreditation to:
- raise awareness of quality issues; and
- encourage organisation-wide commitment to quality assurance.
Promoting Health and Disability Standards
2.24
The Ministry has promoted the preparation and publication of standards under the Health and Disability Services (Safety) Act 2001. To date, the Minister has approved four:
- Health and Disability Sector Standards (NZS 8134:2001);
- National Mental Health Sector Standard (NZS 8143:2001);
- Restraint Minimization and Safe Practice (NZS 8141:2001); and
- Infection Control (NZS 8142:2000).
2.25
The Health and Disability Sector Standards are central to the quality and risk management framework for the delivery of health care services. Compliance with those standards will underpin accountability for DHB performance, and service providers will have to meet the specified standards in order to be certified.
2.26
Infection control management and related risk management activities – such as the safe administration of medicines, cleaning, and laundry services, and the disposal of waste and hazardous substances – are all identified as contributing to safe and effective management of health care services.35
2.27
The Infection Control Standard was published by Standards New Zealand in August 2000 in anticipation of the Health and Disability Services (Safety) Act 2001. It contains valuable information on principles and systems that form the foundation for effective infection control. However, it is not intended to provide a detailed manual of guidance on the Standard’s application. We consider such guidance in Part Six of our report.
2.28
Health care service providers – including public hospitals – will have to demonstrate that they meet the Standard in order to be certified by 1 October 2004. The Standard is accompanied by an infection control audit work-book that has two purposes:
- to establish whether the service provider has attained the outcomes required by the Standard; and
- to identify areas of compliance, and those areas that need to be improved in order to reduce risk levels and provide safe services to patients.
2.29
In anticipation of the quality requirements about to be introduced by the Health and Disability Services (Safety) Act in 2001, the Ministry undertook a clinical survey of hospital services. The Ministry’s survey reviewed the progress hospitals had made towards implementing the Standard – in particular, the implementation of antibiotic policies. The results indicated that some DHBs needed to improve their policies for the use of antibiotics, and their procedures for evaluating compliance with these policies. DHBs were encouraged to achieve compliance by June 2002.
Conclusions
2.30
The two main pieces of legislation that are relevant to the management of hospital-acquired infection provide a sound framework that supports:
- accountability for the quality of health care; and
- the application of principles and systems necessary for quality assurance.
2.31
The national standards-based certification regime required under the Health and Disability Services (Safety) Act 2001 is a potentially effective model if it is properly applied. The Ministry has taken useful steps to facilitate the transition to certification – notably by promoting voluntary accreditation among health care service providers, including public hospitals.
2.32
The Ministry and Standards New Zealand have made good progress in providing information to help hospital services work towards meeting the Standard. The Standard addresses the basic principles and systems that are the foundation of effective infection control. To be certified, hospital services must meet the Standard.
2.33
The Ministry has drawn up a number of supporting strategies to guide the health and disability sector in giving effect to The New Zealand Health Strategy. For example, the infectious disease strategy defines objectives and ways to reduce the incidence of infectious diseases, and action plans to improve the prevention and control of hospital-acquired infection.
Using Expert Advisory Groups
2.34
The Ministry uses (and is a member of) advisory groups that bring together sector knowledge and expertise, and provide technical and scientific advice to the Director-General of Health.
2.35
In respect of infection control, the Ministry has two standing advisory groups – the Infectious Diseases Advisory Committee and the Antibiotic Resistance Working Group. Established in 1996 and 1998 respectively, their roles are to advise on communicable diseases and antibiotic resistance.
2.36
Minutes of meetings of these groups indicate that they have played a valuable role in advising the Ministry on a wide range of policy and operational matters. However, the minutes also illustrate that administration of the groups could be improved.
2.37
In the course of our DHB visits some group members raised concerns about administration matters that could easily be rectified:
- meetings were sporadic and/or infrequent;
- members received material for the meetings with insufficient time to read and prepare; and
- meeting minutes were distributed late, limiting their usefulness to members as a meeting record.
2.38
From time to time, the Ministry also sets up groups to work on issues of national significance. One such example is the working group that drew up the Ministry’s Guidelines for the Control of Methicillin-resistant Staphylococcus aureus (MRSA) in New Zealand, which were published in August 2002. The group comprised medical, laboratory, and infection control personnel.
2.39
The Guidelines contain comprehensive, practical advice, including recommended screening and precautionary practices, covering the various dimensions of MRSA prevention and control. They provide valuable guidance to health care practitioners in drawing up MRSA policies at a local level.
Conclusion
2.40
The Ministry convenes and makes good use of expert advisory groups to advise on a wide range of policy and operational matters. There is scope to improve the way the groups operate.
| Recommendation 1 |
|---|
| The Ministry should review the administration of its expert advisory groups relating to infection control, to enable the groups to be fully effective. |
Undertaking National Surveillance
2.41
Planning, implementation, and evaluation of public health practices require good quality health information. Surveillance refers to the ongoing systematic collection, analysis, and interpretation of health data needed to provide such information.
2.42
Comprehensive national surveillance of hospital-acquired infection would involve:
- promoting agreement among DHBs on data to be collected, and on reliable methods for collecting consistent data to support valid comparisons;
- interpreting the data against factors such as hospital size and service complexity so that meaningful comparisons can be drawn;
- disseminating the results of the analysis to DHBs so that they can compare their performance;
- using the information to provide assurance to the public on matters relating to hospital safety;
- identifying unexpected results that might indicate weaknesses in infection control practices that could be addressed through guidelines; and
- monitoring trends in order to gain an early warning of possible epidemics caused by infectious organisms.
2.43
Two types of national surveillance currently undertaken are relevant to hospital-acquired infection:
- surveillance of organisms of public health significance; and
- collection and reporting of hospital-acquired bloodstream infection rates.
Surveillance of Organisms of Public Health Significance
2.44
Surveillance of organisms of public health significance is carried out by the Institute of Environmental Science and Research Limited (ESR), a Crown Research Institute specialising in environmental and public health, and forensic science. ESR provides a range of laboratory services for such things as food safety, pharmaceuticals, and communicable diseases. For communicable diseases, it provides laboratory services to other laboratories in hospitals and other organisations, and assistance to public health units within DHBs in managing outbreaks of infection in the community.
2.45
Under contract to the Ministry, ESR examines and reports on organisms of public health significance referred to it by medical laboratories throughout the country. All medical laboratories are required to send specified isolates36 to ESR for further testing. Among these specified isolates are a variety of antibiotic-resistant bacteria that are common causes of hospital-acquired infection.
2.46
ESR operates national surveillance systems, including notifiable disease surveillance and specific non-notifiable disease surveillance (e.g. for sexually transmitted infections). The systems are wide-ranging and are concerned with:
- hazard – laboratory surveillance of organisms, such as MRSA;
- exposure – surveillance of transmission routes for organisms, such as food or water; and
- outcome – surveillance of the disease, based on the organism, such as cases of meningococcal disease.
2.47
ESR’s laboratory-based surveillance includes confirmation and investigation of important and emerging antibiotic-resistant organisms. These include MRSA which, among multi-drug-resistant organisms, is the most common cause of hospital-acquired infection. MRSA isolates sent to ESR are accompanied by information about the circumstances surrounding the discovery of the infectious organism. In 2000, hospital and community laboratories referred isolates of multi-drug-resistant MRSA from 1003 people, the majority of whom were hospital patients.
2.48
Recorded information about the circumstances surrounding the acquisition and transmission of MRSA includes:
- details of the patient, including whether they have been previously admitted to hospital in New Zealand or overseas;
- the source and nature of the organism – whether the site from which the isolate was taken was infected and whether the MRSA strain is resistant to antibiotics; and
- the nature and extent of staff contact with the organism, the site from which the specimen was taken, and whether and when the staff involved have been screened.
2.49
ESR publishes a weekly update on reported incidences of MRSA. Each month it also publishes data related to MRSA and other organisms of public health significance in the monthly Public Health Update.
National Surveillance of Hospital-acquired Bloodstream Infections
2.50
In the November 2001 strategy An Integrated Approach to Infectious Disease (see paragraph 2.10) the Ministry stated its intention to:
… establish a national surveillance programme for infections acquired in health and disability care institutions, including standardised definitions and data analysis, with local feedback mechanisms to clinicians.
2.51
This objective was one of a number of high-priority initiatives for which funding has not yet been identified, and which must be assessed alongside other priorities. The Ministry did not set a timetable for establishing this national surveillance programme.
2.52
A national system was (and still is) in place for monitoring one type of hospital-acquired infection – bloodstream infections. The system was set up in 1994.
2.53
Trends in bloodstream infection rates are an important measure of DHB performance in delivering good quality health care services because:
- While bloodstream infections comprise only 5% to 10% of hospital-acquired infections, they can have very high mortality rates – higher than 30%. Reducing bloodstream infections can therefore save lives.
- Patients with hospital-acquired bloodstream infections tend to stay in hospital longer and cost more to treat.
- High levels of such infections can also indicate shortcomings in the quality of patient care, particularly in relation to the use of intravenous catheters.
2.54
The Ministry collects quarterly data on hospital-acquired bloodstream infections from all DHBs as a measure in the Balanced Scorecard37 reports that form the basis for its Quarterly Review of Comparative Hospital and Health Service Performance for the Minister of Health. The rate of bloodstream infection (determined by the number of hospital-acquired blood-stream infections during the quarter divided by the total number of inpatients38 during the quarter, and expressed as a rate per 1000 inpatients) is one of four indicators for quality of service and patient satisfaction.39
2.55
The quarterly reports to the Minister contain comments on trends in rates of hospital-acquired bloodstream infection and on possible reasons for the trends. This analysis is not made available to DHBs on a regular basis, but could be useful to them in reviewing their results (subject to the concerns about data quality explained in paragraph 2.61).
2.56
We asked senior hospital managers in the DHBs how useful they found the Ministry’s collection and reporting of rates of hospital-acquired bloodstream infection. We also asked infection control teams about their use of bloodstream infection data.
2.57
Hospital managers had mixed views on the usefulness of bloodstream infection reporting to the Ministry. Between 70% and 80% of hospital-acquired bloodstream infections occur in the six DHBs with tertiary hospitals (see Figure 1 on page 33). Because of the higher level of such infections (and the associated costs) in the larger DHBs, they should find the bloodstream infection reporting particularly useful. However, only two of the six DHB tertiary hospital managers said that they found the reporting useful.
2.58
The results indicate that some DHB managers may not be aware of the potential benefits from using this data as an indicator of quality, or as a tool to compare their performance with that of other DHBs, and to identify possible risks and scope for improvements.
2.59
More than two-thirds of DHB infection control teams did not find the reporting useful, and noted that it could be improved by:
- compiling comparative data which would allow them to share information and benchmark their results against those of other DHBs;
- incorporating more detailed data which would help identify trends, resistance patterns, and risk factors (such as case mix and significant organisms);
- adopting a definition of bloodstream infection which took account of the severity of the infection and the circumstances in which such infection occurred; and
- more timely reporting back to DHBs.
| "… Ministry of Health balanced scorecard reports give details of our results only and are of little value …" |
2.60
We followed up with the Ministry the suggestion that it compile data for benchmarking, since we understood that DHBs were already receiving some comparative data from the Ministry. The Ministry confirmed that it provided comparative information to DHBs.
2.61
In addition, the Ministry told us that it considered the bloodstream infection data it received from DHBs to be unreliable, because of inconsistencies in the way DHBs defined such infections.
Conclusions
2.62
The Ministry has made a commitment to establish a national surveillance programme for hospital-acquired infection, but has not yet set a timetable for meeting this commitment. As a result, it is not currently possible to provide assurance to the public on related hospital safety matters, and DHBs cannot comprehensively compare their performance.
2.63
The wide-ranging national surveillance that ESR undertakes in other areas illustrates its experience in designing, establishing and operating national surveillance, and in the analysis, reporting, and dissemination of surveillance data. Its current work in relation to hospital-acquired infection (under contract to the Ministry) is primarily concerned with scientific analysis of selected organisms. It is therefore not designed to meet the requirements of a comprehensive national surveillance programme, although it meets some of these requirements in respect of MRSA.
2.64
There is a national system for monitoring one important, high-risk sub-set of hospital-acquired infection – bloodstream infections. However, the data collected is neither reliable nor consistent among DHBs, and in some DHBs comparative data may not be reaching those people who can make best use of it. The monitoring is of limited value to DHBs wanting to use it to help them improve their management of hospital-acquired infection.
| Recommendation 2 |
|---|
| In consultation with DHBs, the Ministry should review the way it collects and feeds back data on bloodstream infections, to make such data more reliable and consistently available. |
| Recommendation 3 |
|---|
| The Ministry should work with ESR and DHBs to establish a timetable and project plan for setting up national surveillance of hospital-acquired infection along the lines indicated in paragraph 2.42 on page 49. |
28: The New Zealand Health Strategy; Ministry of Health, Wellington, December 2000; ISBN 0-478-23993- 9 (Book), ISBN 0-478-23994-7 (Internet) www.moh.govt.nz/nzhs.html.
29: Chapter 3 of The New Zealand Health Strategy.
30: Page 11 of The New Zealand Health Strategy.
31: Appendix 6, Provider Quality Specifications – Operational Policy Framework 2002-03, Ministry of Health.
32: Section 3 of the Health and Disability Services (Safety) Act 2001.
33: Director-General is defined as the chief executive of the Ministry under the State Sector Act 1988.
34: Section 21 of the Health and Disability Services (Safety) Act 2001.
35: Part 5 of NZS 8134:2001 Health and Disability Sector Standards.
36: An isolate is an organism recovered from a specimen and grown in culture media.
37: The Balanced Scorecard reports four areas of DHB performance: quality of service and patient satisfaction; organisational health; process and efficiency; and financial performance.
38: An inpatient is a patient admitted for treatment and present at the midnight census (excluding well babies and boarders).
39: The other three areas are: patient satisfaction, complaints resolved in 30 days, and the time taken to assess emergency patients.
page top